Table of Contents (click to expand)
Gold has extraterrestrial origins. It was formed as a result of complex chemical reactions from a cataclysmic celestial explosion.
Gold is one of the most sought after metals on Earth. It has been prized for its beauty, malleability and non-corrosive nature for thousands of years.
It has also played an important role in numerous ancient civilizations. The Egyptians called gold the “breath of God.” Gold also finds mention in Greek mythology, where King Midas was given the boon of a “golden touch” by Dionysus for saving his life. Midas could turn anything to gold by mere touch.

Gold is typically buried deep inside the earth’s crust. We have discovered about 244,000 metric tons of gold, of which 187,000 tons have been mined. China, Australia and South Africa are the three major gold-producing countries. (Source)
But how exactly did we end up in these gold mines?
Origins Of Gold
Gold in its pure form is not ‘made’; it occurs naturally deep within the earth. The Incan civilization believed that gold was “the sweat of the sun”. Though it was a superstitious belief, they got one thing right—gold does originate from a star!

Thanks to nuclear fusion, the Sun can create many elements, including iron, nickel, calcium, sodium, etc., along with elements as heavy as uranium. Nevertheless, it does not generate enough energy to synthesize gold. Gold formation is only possible when a star explodes in a supernova, pumping out massive amounts of energy.
An alternative theory suggests that the collision of neutron stars can enable the formation of gold.
Also Read: Can Giant Stars Fuse To Form Gold?
Gold Is Extraterrestrial
Cataclysmic celestial explosions, i.e.. supernovae, are responsible for the abundant supply of gold on Earth. Stars are mainly composed of hydrogen, but when a star accumulates too much matter, it collapses in on itself due to the pressure from its gravitational pull.
As the dying star expands into a red giant, hydrogen transforms into helium. As the helium atoms fuse, it leads to the formation of carbon, followed by oxygen.
Along with energy, a star produces a large number of neutrons during a supernova. These neutrons fuse to form heavier elements like iron and copper.
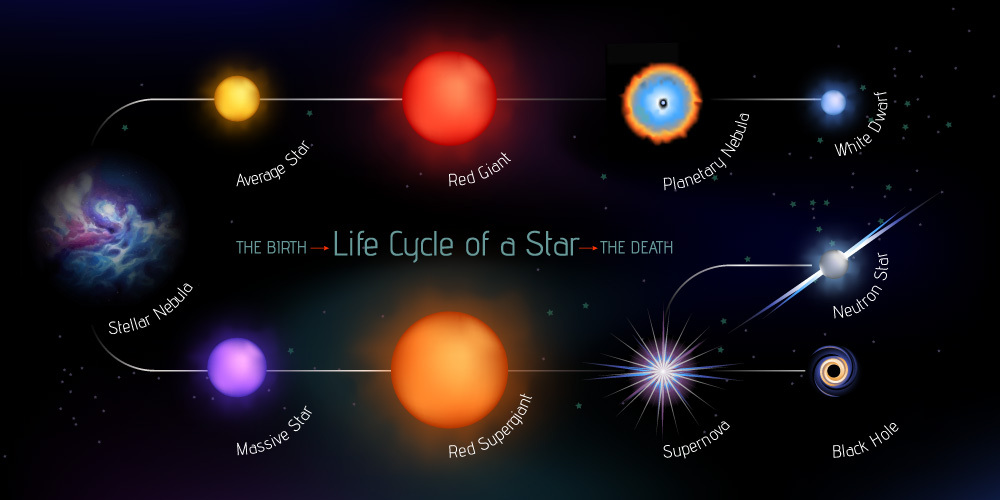
However, under extreme conditions, the exploding star can produce heavy metals like gold and uranium through a process known as the rapid neutron capture or r-process.
Here, an atom swells to a very unstable size as the neutrons released during the supernova bombard its nucleus. Due to this, the formation of the heaviest elements – gold & uranium – takes place within seconds. The whole process happens so fast that the unstable element doesn’t have the time to split apart.
As the dying star explodes as its last hurrah, it sends out shockwaves, propelling mineral-rich debris throughout space.
This tells us where and how the formation of gold occurs, but what explains its abundant presence on Earth?
Gold Hitched A Ride To Earth With Meteors
The gold formed out in the universe ended up deep inside the earth’s core. The only logical explanation for the arrival of an extraterrestrial element on Earth is that… it might have taken a lift from meteoroids.
Billions of years ago, during the initial formation of Earth, heavy elements like iron sank into the center of the earth to form its core.
If gold had come here when the earth was still forming its core, then the molten iron would have dragged other heavy elements, such as the gold, along with it to the core. However, we can easily find gold deposits in the earth’s crust.
A study conducted by the University of Bristol gives us insights into the period when gold sunk to the earth’s core. They studied the isotope concentration of rocks from different ages, which gives evidence that gold landed on Earth after the formation of the planet’s core.
Earth’s mantle saw a change in its composition after the planet was hit by meteors 3.8 billion years ago. These meteors also brought with them loads of precious gold. The bombardment lined the planet’s crust and mantle with a thick layer of gold.

Turns out, the gold that we lavish today has a very ‘explosive’ history. Life on Earth had to suffer the assault of meteors weighing billions of kilos just for us to enjoy some of this fancy gold.
Is It Possible To Synthesize Gold On Earth?

Many alchemists in the past have tried reacting different chemicals together with the hope of making gold. In principle, yes, we can create gold from other elements, but the process of creating gold would cost way more than it’s worth.
The atomic number of Gold (Au) is 79, so by default, any element containing 79 protons will be gold. In theory, gold can be created by adding or removing protons from copper (Cu) and mercury (Hg), respectively. The removal or addition of protons to an atom is a form of a nuclear reaction, and as you may have guessed, it’s an expensive job to conduct a nuclear reaction.
Many alchemists have tried and failed in their endeavors to create gold from other elements. Some scientists even tried turning lead into gold!
To learn how that turned out, check out this article.
Also Read: Can We Transform Lead Into Gold?
Conclusion
From being an integral part of jewelry to being used as a garnish for some delicacies, we use gold all over the world for various purposes.
Many might assume that, like any other element, chemical reactions within our planet might cause gold formation, but as scientists have found out, gold is an alien element. After exploding out of a dying star, it burst through our atmosphere riding on meteors to bedazzle our lives!
How well do you understand the article above!

References (click to expand)
- How is gold made? - UCSB Science Line. The University of California, Santa Barbara
- Willbold, M., Elliott, T., & Moorbath, S. (2011, September). The tungsten isotopic composition of the Earth’s mantle before the terminal bombardment. Nature. Springer Science and Business Media LLC.
- Croswell, K. (2021, January 20). Tracing gold’s cosmic origin story. Proceedings of the National Academy of Sciences. Proceedings of the National Academy of Sciences.
- Can gold be created from other elements?. West Texas A&M University
- Meteorites Delivered Earth's Mineable Gold - Scientific American. Scientific American
